Exton Company’s Technology Helps Wean COVID-19 Patients Off Ventilators
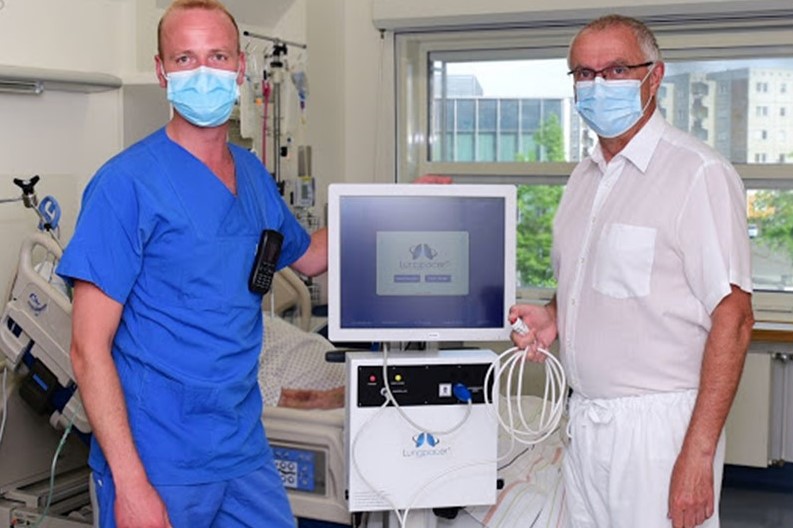
Lungpacer Medical, a Canadian medical device company with U.S. operations in Exton, has developed technology that is being used to wean COVID-19 patients off ventilators, writes John George for the Philadelphia Business Journal.
The FDA awarded the company’s Lungpacer system with emergency use authorization in April. The portable device features a catheter that is used to deliver fluids and medicine to stimulate the weakened muscles of the diaphragm.
“As soon as the pandemic became prevalent in the United States, we saw a lot of patients were requiring extended ventilator assistance,” said Doug Evans, CEO of Lungpacer Medical. “We started a dialogue with the FDA about our device, and they encouraged us to apply for emergency access.”
So far, fewer than 10 people have been treated with the Lungpacer system, but the company is now meeting with hospitals and providing them with educational materials to increase awareness of the technology.
The company is also conducting trials in several countries and is expecting to use the data when seeking regulatory clearance from the FDA in 2022.
Read more about Lungpacer Medical in the Philadelphia Business Journal here.
You Might Also Like
Paoli’s Audacity Therapeutics Aims to Raise $150 Million to Test if SARS Drugs Can Treat COVID-19
After His Father Was Hospitalized with COVID-19, Local Man Made Sure Their Exton Business Survived
Chester County Hospital Celebrates as Local Woman Beats COVID-19 After Three Weeks in ICU
Connect With Your Community
Subscribe to stay informed!
"*" indicates required fields






























![95000-1023_ACJ_BannerAd[1]](https://vista.today/wp-content/uploads/2023/03/95000-1023_ACJ_BannerAd1.jpg)

















